Gamma Rays… The most well-known type of nuclear radiation, thanks to the efforts of comic books and movies for the past several decades. However, unlike comic books, being exposed to gamma radiation will not actually turn you into a giant green rage monster, and also unlike what anti-nuclear advocates have tried to tell people, it won’t even turn you into a single giant tumour the same size as The Incredible Hulk.
What gamma radiation is is actually very simple. It is nothing more than a photon that has a very short wavelength. Not actually much different from the same photons that allow us to see the colours of the flowers in the garden, or any other lovely sight that we might take for granted on a daily basis.
Unlike alpha or beta or spontaneous fission, gamma radiation emission does not cause transmutation. Thus a gamma emitting element will not change into another element after releasing its gamma ray. This is probably the single most important thing to know about radiation. A single atom of a gamma emitting element will generally only ever release one gamma-ray photon until/unless it then undergoes another alpha, beta or neutron decay and becomes a different gamma emitting element. This is very different from almost every portrayal of gamma radiation in public circulation I can think of, where it is made out to be some kind of continuous emission in perpetuity, like a lightbulb left on forever. But if you think way back to your jr. High school physics class, that pesky Second Law of Thermodynamics. “The entropy of an isolated system will always trend towards a maximum.” Paraphrased by science fiction author Larry Niven as “There’s no such thing as a free lunch”, or more commonly as “you can’t get something from nothing.”
“But what makes an element emit gamma rays?” I hear the astute among you ask? What gives an atom the energy to create and release a gamma ray? The answer is, of course, nuclear decay. In some atoms, after an alpha, beta or spontaneous fission occurs, the daughter product atom(s) are left in an excited state and to calm down the nucleus releases a gamma ray photon or two in order to relax to it’s lowest energy state. Thus it is best to think of a gamma ray as the younger sibling of alpha and beta radiation, always wanting to tag along and running around much faster than their tired older sibling.

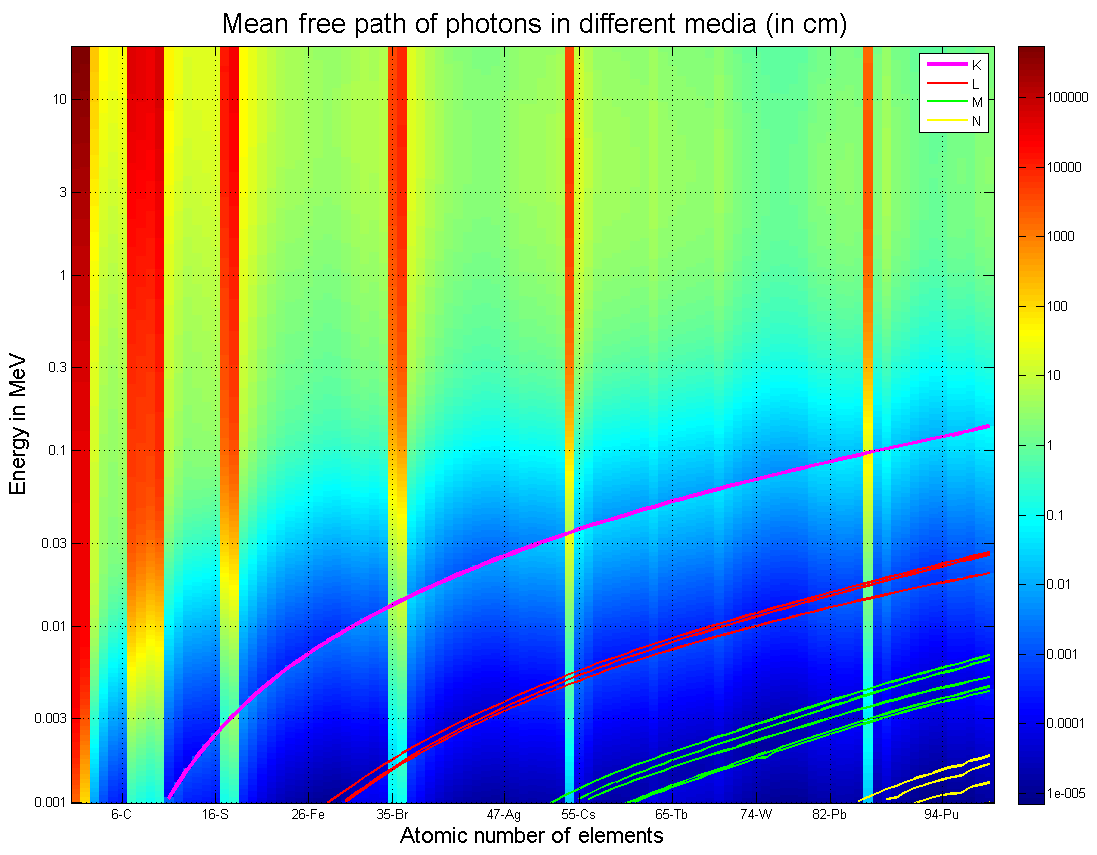
Much faster is a bit of an understatement, since gamma rays are just very high energy photons, they literally travels at the speed of light. This makes it quite difficult to stop them inside of a containment area. Gamma rays have to be treated more seriously than alpha or beta particles because they are uncharged, small, and literally as fast as it is possible to be. Thus, it takes significantly more shielding to stop them than it does for any of the types of radiation. But luckily even though it takes more material to stop them they are still not as damaging as neutron radiation as gamma rays do not leave behind residual radiation after the initial exposure.
To protect against gamma-rays is quite simple if a touch cumbersome. You just require thick sheets of the densest material you can afford to buy, which in most cases is usually lead although in some specific structural cases it could be something like tungsten which is significantly stronger.
There is a great deal of math, ranging from mid-level to exceptionally sophisticated that describes how to properly shield from gamma rays, but what it all boils down to is that the density of the material is king. Lead is about 11.36 g/cm^3 ( 0.410 lb/in^3 for you Freedom unit users) and it will stop approximately 50% of all gamma rays that attempt to pass through it for every centimeter of thickness of the lead. So 2 cm will stop 75%, 3 cm will stop 87.5%, 4cm stops 93.75% and so on.
It is possible to create shielding that reduces the measurable gamma exposure outside of a reactor to 1 billionth of what it is inside the reactor. That means only one gamma ray out of every billion is allowed to sneak through. Shielding of this level of effectiveness requires approximately 39-40 cm (1.3 feet) of lead. If you are using water instead of lead, as in the case of a storage pool, the same amount of shielding requires approximately 4.2m (13.8 ft) and concrete would require 2 m (6.6 ft). Again remember that gamma radiation doesn’t make other materials radioactive, thus the water used in storage pools is perfectly safe, regardless of what people who are trying to scare you about nuclear power say.
Gamma rays seem very scary, but in fact they are amazingly predictable, which makes them easily defended against. Scientists have found more than two thousand unique gamma decay steps across every known radioactive element. We also know how long it takes for the element to release the gamma-ray and how much energy that gamma-ray contains. Don’t believe me? Take a look at this table then.
Now, if that doesn’t make your head spin I would draw your attention to arguably the most important part of that table, the column of Intensities. they are defined as gamma emissions per 100 parent atom decays. what this column shows is that in the vast majority of cases, you will get less than one gamma ray from every hundred other decays. so it’s not like Every decay of every element ever releases gamma rays. in fact, they are generally quite rare compared to alpha and beta decays. So unless you purposefully ignore a lot of very large, very eye-catching safety signs, you don’t have to worry about gamma-rays.
Not only that but since your body is made mostly of water, you as a human being are actually quite bad at stopping gamma rays. in fact on average a gamma ray passing through you from top to bottom will probably interact with fewer than 100 atoms in your body.
Now I’m not going to say that gamma rays can’t be dangerous, because everything can be dangerous if you have too much of of it. Gamma rays, fire, water, alcohol, oxygen, string… it’s all in how you handle the risks in order to benefit yourself and others. And when the benefits are as far reaching as they are with nuclear power, it’s important to know what the actual risks are so that you aren’t scared away from it for no reason.
